CoP Containment
About
In ISPE D/A/CH, members are actively involved in regional working groups and the CoP’s (Communities of Practice). Through technical/topic-based workshops, discussions, and communication, best practice working papers and approaches are developed in the sensitive environment of patient safety. They serve as guidelines for the industry and the authorities. Members of ISPE D/A/CH participate in the ISPE Guides.
Introduction
Containment
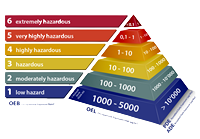
The topic of containment moves the pharmaceutical industry. Many newly introduced drugs are effective even in small doses – their ingredients are highly active. For patients, this development is an opportunity; for manufacturers, it is a challenge. Low occupational exposure limits and complex toxicological assessments require special measures for process safety in the laboratory and in production.
The new Containment Manual
Now available – the new edition of the successful Containment Handbook of the Working Group.
The German Containment Handbook in the 2nd edition is available as a book.
The manual covers risk assessment, containment life cycle, process requirements, technical systems, validation, cleaning and personnel. Each chapter of the handbook begins with a definition or introduction, concepts, processes, technologies are identified and described in the form of best practices or as illustrated examples. The new EMA guidelines, “Guideline on setting health based exposure limits for use in risk identification in the manufacture of different medicinal products in shared facilities”, are also taken into account.


Lead CoP Containment
Dr. Martin Schöler
Fette Compacting GmbH

Co-lead
Oliver Gottlieb
PharmaCon Service ApS

Secretary
Jessica Redmer
AiCuris Anti-infective Cures AG
Steering Committee
Hristina Freitag,
Vetter Pharma-Fertigungs GmbH & Co KG
Dirk Collins,
WALDNER Process & Automation Solutions (PAS)
Michael Maintok,
Glatt GmbH
Dr. Patrick Sproll,
Lonza AG
Uwe Hahmann,
HET Filter Ltd.
Daniela Kovats,
Fareva S.A.
Dirk Daegele,
Vetter Pharma-Fertigung GmbH & Co. KG
Michael Donner,
IPS-Integrated Project Services GmbH
Reiner Lemperle,
HECHT Technologie GmbH
Dr. Andreas Schreiner,
Novartis Phama AG
Bernhard Steidle,
Boehringer Ingelheim Pharma GmbH & Co. KG
Christopher Pfanstiel,
Syntegon Technology GmbH
Dr. Rainer Nicolai
F. Hoffmann – La Roche AG
Birger Bockius
Merck Electronics KGaA
Dr. Reinhold Maeck,
Boehringer-Ingelheim Corp. Center GmbH
Tim Luebke,
Bayer AG
Lukas Jürgen,
Boehringer-Ingelheim Corp. Center GmbH